The Need for New Therapies for Type 1 Diabetes
Original Article: https://www.medicalnewstoday.com/articles/fda-approves-first-cellular-therapy-for-type-1-diabetes-what-does-it-do
In 2021, about 8.4 million people globally were living with type 1 diabetes (T1D). There is currently no cure and those with T1D must use insulin for their entire life. For some individuals their life can be significantly harmed by severe hypoglycemia (very low blood glucose levels leading to dizziness, confusions, loss of consciousness, or coma) The United States Food and Drug Administration (FDA) recently approved a new therapy for T1D called Lantidra.
How does it work?
Lantidra is made from a special method for obtaining pancreatic cells from deceased donors. These cells, known as allogeneic islet beta cells, have the ability to make insulin and put it into the bloodstream. Lantidra has been approved for the treatment of adults with T1D who suffer from many episodes of severe hypoglycemia despite careful diabetes management and education.
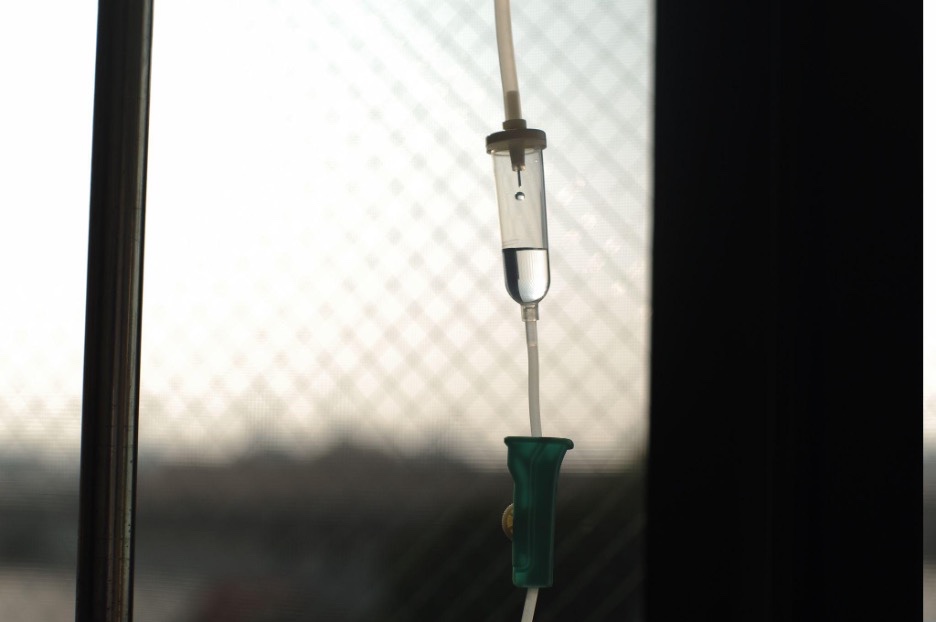
First Pancreatic Islet Cell Therapy
With this new therapy, healthy insulin producing cells are given from an IV line into the main vein of the liver of the person with diabetes. It is important to note that anti-rejection medications are needed to stop the transplanted islet cells from being destroyed by a person’s own immune system.
The transplanted beta cells produce all the insulin that a person needs for regulating blood glucose levels without the need for additional insulin injections or pumps.This is similar to islet cell transplantation which has been available as a research treatment.
What did the research show?
In a clinical study of 30 adults with T1D and hypoglycemia unawareness, 21 participants did not have to use insulin for one year or longer. Among them, 10 participants remained insulin-free for more than five years. However, it is important to note that not all participants experienced the same level of success, and some did not achieve any days of being able to go without insulin shots.
Are there any concerns about Lantidra?
So far, side effects from the infusion and from the anti-rejection drugs can occur and some have been serious. Of the 21 study participants, 90% experienced at least one serious adverse reaction related to the procedure and the medications. More research is needed to balance the risks and benefits for the wider community of people living with T1D.

